Isotopes of chlorine 35 and 378/15/2023 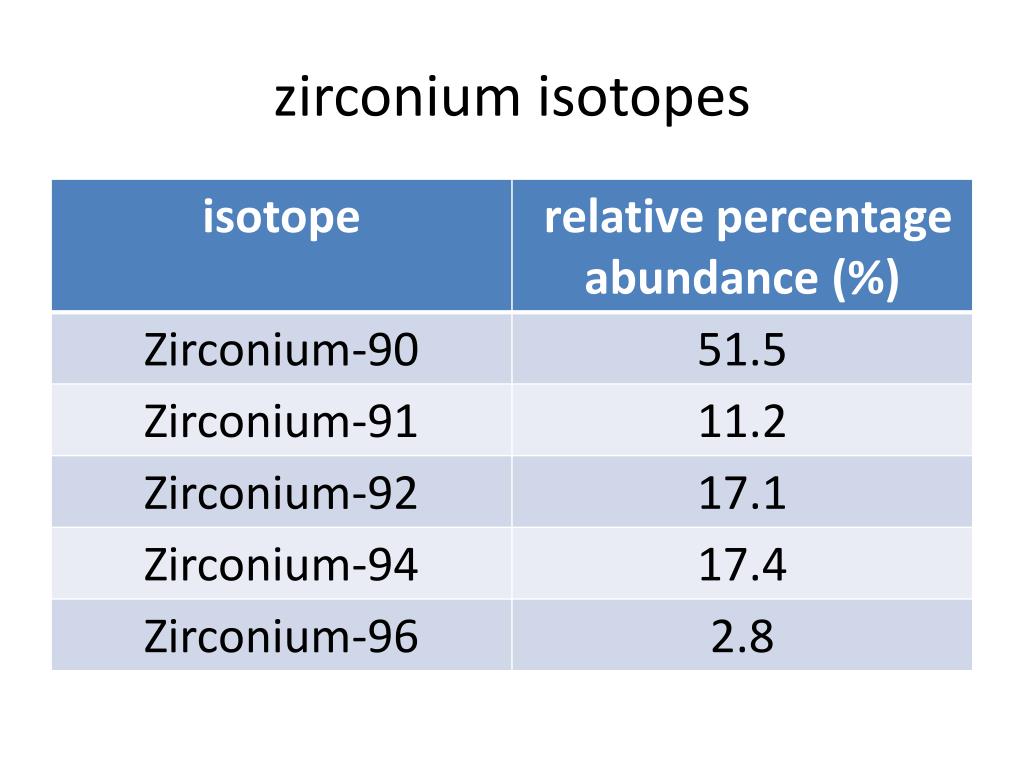
Neutron number plus atomic number equals atomic mass number: N+ZA. The total number of neutrons in the nucleus of an atom is called the neutron number of the atom and is given the symbol N. I know that in 100 atoms, 75.53 of those atoms would be chlorine-35 and 24.47 of those atoms would be chlorine-37. Mass numbers of typical isotopes of Chlorine are 35 37. If I wanted to determine the mass of 100 atoms of chlorine, I would need to know how many of each isotope of chlorine are in 100 atoms. The easiest way to do this is to pretend that I had 100 atoms of chlorine. Because these percentages are not 50/50, the percents must be figured into the individual mass of each isotope to determine the average mass of the element. A little less than 25% of the chlorine is chlorine-37. According to the percent abundance data, over 75% of all the chlorine that exists on this planet is of isotope chlorine-35. This discrepancy is due to the abundance of chlorine on this planet. Solution Verified by Toppr Correct option is C) Was this answer helpful 0 0 Similar questions If an element z having atomic weight x exists in two isotopes of mass number (x1) and (x+2) then, the percentage abundance of heavier isotopes is: Hard View solution > 1735Cl and 1737Cl are two isotopes of chlorine. The average atomic mass for chlorine is 35.453. 
You will notice that the average atomic weight for chlorine on the periodic table is not 36 (the average of the numbers 35 & 37). For example, there are two naturally occurring isotopes of chlorine.ĭetermining the Atomic Mass from Isotope Data The difference between chlorine-35 and chlorine-37 is that chlorine-35 has 18 neurons, while chlorine-37 has 19 neurons. The isotopes of chlorine Cl-35 and Cl-37 should be placed in the same slot because their chemical properties are the same. 30 The isotopes of chlorine with mass number 35 and 37 exist in ratio if its average atomic mass is 35.5 of. The mass number is the sum of the number of protons and neutrons for that isotope. To identify the different isotopes, scientist add a mass number after the element’s name. This fact that you can have atoms of the same element with different masses is what we call isotopes. The only difference is the number of neutrons, which affects the mass of the atom. The natural abundance of chlorine-35 is (greater than. Each atom is hydrogen, and is identical in chemical properties. Chlorine has two common isotopes, chlorine-35 and chlorine-37, and an atomic mass of 35.45 amu. Hydrogen-3 has a mass of 3 (1 proton and 2 neutrons). Hydrogen 2 has a mass of 2 (1 proton and 1 neutron). Hydrogen-1 has a mass of 1 (1 proton and 0 neutrons). Hydrogen has 3 naturally occurring isotopes. 
Therefore, the only difference between the atoms are their masses. Mass numbers of typical isotopes of Chlorine are 35 37. The only difference is the number of neutrons within the nucleus. The chemical properties of each isotope are identical because they are the same element. Isotopes of Chlorine with mass number 35 and mass number 37 exist in the ratio 3 : 1 Explanation: The molar mass of Chlorine is 35.5 Let the mole fraction of isotope 35 be x and that of isotope 37 be (1 - x). Elements that are of the same kind (i.e., 2 carbon atoms) that have different masses, are called isotopes. However, like the electrons, the amount of neutrons in any atom of that element can be different. These two are the most stable isotopes of chlorine atoms.Every atom of a given element has the same number of protons. Let take percentage abundance of $^$, therefore we take the average of all chlorine atoms. We know that isotopes are defined as the species that are one of two or more species of atoms of an element which have the same atomic number and position in the periodic table but different atomic masses. Let suppose any atom has two isotopes then the formula of average relative atomic mass of atom is Average relative atomic mass of a atom= /$100$. Hint : We will solve this problem with the help of the formula of finding the average relative atomic mass of an atom when atomic masses of their isotopes are known.
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
